
Elements with high electronegativity will want to take electrons from other atoms, whereas elements with low electronegativity will easily give their electrons away.Ĥ. Fluorine is the most electronegative element while francium is the least.ģ. The trend for electronegativity points from Fr, francium, to F, flourine. Electronegativity is a property of an atom that describes how much it wants electrons.Ģ. By doing so, both sodium and chloride satisfy their octets.Įlectronegativity is a very important concept in chemistry. The sodium atom gives away one electron from its valence shell to the chlorine atom, which is missing one valence electron. Later on, we'll use the concept of electronegativity to explain bonding, or why certain elements pair together. Na can then give one electron to Cl to satisfy both their octet rules. This explains why compounds like NaCl exist in such abundance: the Cl atom, being highly electronegative, really wants an electron whereas the Na atom, being low in electronegative character, really wants to give an electron away. Elements with low electronegativity, also called electropositive elements, will do the opposite by readily freeing up their valence electrons to be taken. 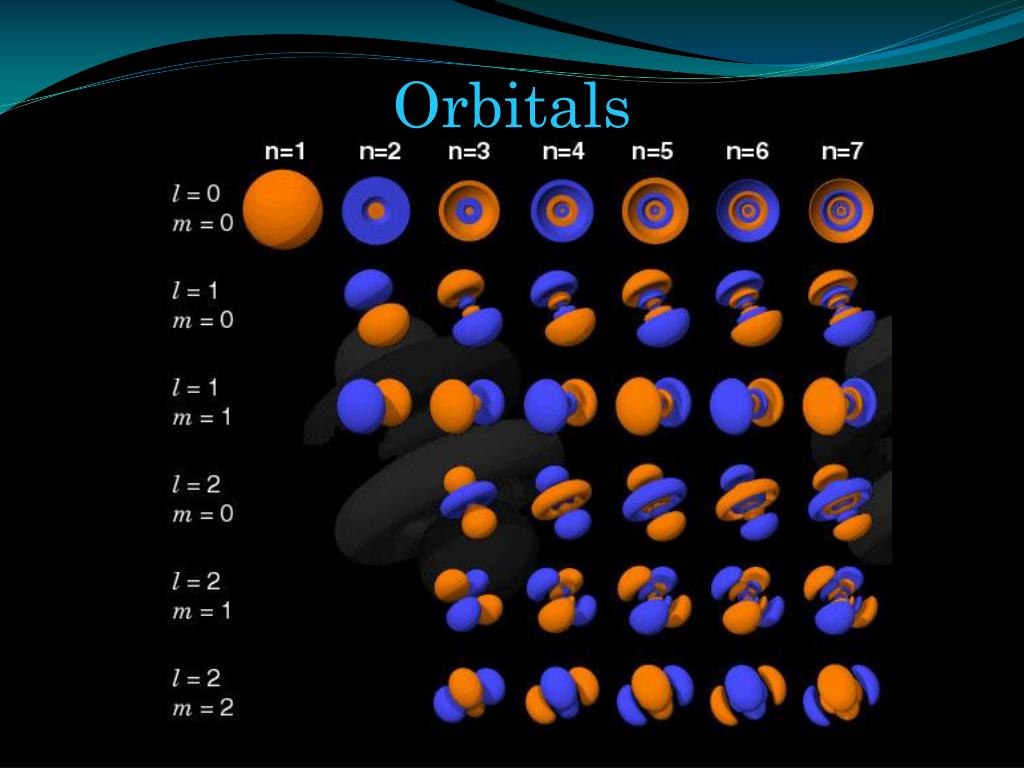
Elements with high electronegativity will hold onto their electrons tightly while searching for more electrons. You can probably see this on the scale itself: the closer an element is to F, the more electronegative it is. If you draw an arrow from Fr to F, you will have described the trend for electronegativity. The trend for electronegativity is as follows:į is the most electronegative element, and Fr the least. The frugal person is the electronegative atom, and the wealthy is the opposite (electropositive).Įlectronegativity has been quantified into what is called the Pauling Scale of Electronegativity:Īs seen on the modified periodic table, group 17 elements have higher electronegativity values than group 1 elements. He really wants to be charitable and give away some of his money (fictional scenario). On the other hand, imagine a wealthy person who has more money than he needs. This person is likely to hold onto all his money while taking money from whereever he can. Imagine that there's a really frugal person who's saving up for a new car. Once an electronegative atom gets hold of an electron, it will not let go. Electronegativity is a measure of how selfish and greedy an element is for electrons. In fact, there's a measure of how much a specific element wants electrons, called electronegativity. It is because of this that they are called noble gases: the noble do not react to the problems of the commons (apparently).Ī large portion of an element's behavior can be predicted solely by its number of valence electrons. The noble gases are not reactive at all because they have already satisfied the octet rule. This is why halogens are so reactive: they desperately want one more electron and will often times break apart the surrounding atoms in order to get it. Group 17 elements have 7 valence electrons and strongly desire one more to satisfy the octet rule, going as far as to rip the electron from another neighboring atom. This "want" to become like the noble gases is why different elements behave differently. 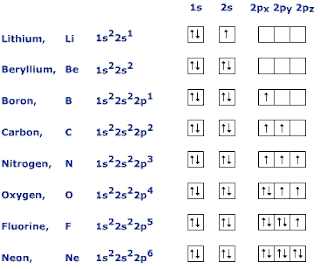
The octet rule can therefore also be stated as follows:Īll elements want to be like noble gases. If you look back at the periodic table, you'll notice that the elements with 8 electrons are in group 18, the noble gases. The octet rule states the following:Īll elements want to have 8 valence electrons. Valence electrons have this interesting behavior which is described by the octet rule. Only the ones in the outermost shell contribute to an element's chemical properties. In other words, the total number of electrons doesn't matter (yet). For elements of the same group, the total number of electrons will be different, but the number of valence electrons will be the same. Similarly, Be has the same number of valence electrons as Mg, Ca, and Sr. F has the same number of valence electrons as Br. The reason behind this is that elements of the same group have the same number of valence electrons. Electrons inside the valence shell are called valence electrons.Īll of the halogens (group 17) are extremely reactive. In the picture above, you can see these different orbitals, labelled `n=1,2,3,4.` These orbitals are called shells and the outermost shell is called the valence shell. In the Bohr Model of the atom, protons and neutrons are in the center of the atom while electrons orbit the nucleus at defined distances, similar to how planets orbit the sun. In the case of atoms, we have positively charged protons and negatively charged electrons. If you think back to the first post, we briefly examined a formula known as Coulomb's Law, which stated that things of opposite charge attract while things of the same charge repel.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
